MAPcore Services
At this time, please contact us for pricing and quotes.
Histology Services
Coring
Coring is a method used to access archival FFPE block samples for use in molecular assays by punching the block from the top downward toward the cassette. Cores can be obtained using 0.6mm, GPEC uses RNAzap for all molecular preparations to preserve nucleic acid samples.
Scrolling
Scrolling, like coring, is a method of obtaining FFPE sample for molecular assays. Unlike coring, scrolling samples the face of the block at the requested thickness using a Leica RM2235 Manual Rotary Microtome. GPEC uses RNAzap for all molecular preparations to preserve nucleic acid samples.
Microtomy
Sectioning at MAPcore/GPEC is done using Leica RM2235 Manual Rotary Microtome. Sections are cut to the requested thickness (typically 4um) and adhered to Leica APEX BOND adhesive microscope slides. Slides can be returned to the requester unstained or processed using below histochemical services.
Microtomy for Macrodissection
Macrodissection is a method of FFPE sample enrichment guided by a pathologist or researcher annotated H&E or IHC stain. Samples are sectioned and the area of interest is isolated by scraping away adjacent tissue.
At MAPcore we will section slides for macrodissection by the investigator.
Tissue Microarray (TMA) Construction
TMAs are powerful tools that allow hundreds of FFPE samples to be analyzed at one time. TMAs constructed at GPEC are done using Pathology Devices TMAarrayer. Available core diameter sizes are 0.6mm, 1.0mm, and 2.0mm. TMA maps are prepared by GPEC based on the number of *donor samples and are given to the researcher for every TMA built.
*Please note, MAPcore and GPEC do NOT provide donor tissue. Tissue must be requested to tumour bank by researcher or directly provided by researchers
Laser Capture Microdissection
Laser microdissection is used for a broad range of molecular assays. The gravity collection method is contact-less to ensure minimal contamination.
Histochemistry Services
Hematoxylin and Eosin
Hematoxylin and Eosin stain at GPEC is done manually using Fisher Scientifics Eosin Y 1% Alcohol and PlatinumLine Hematoxylin Gills 2 Stain.
Single Biomarker DAB IHC
3,3′-Diaminobenzidine immunohistochemistry done at MAPcore/GPEC is fully automated using Leica DAB Refine Detection kits on the Leica Bond Rx system. Our biomarker catalog has undergone rigorous optimization using a range of normal and tumor tissues, positive and negative controls and pathologist evaluation. Custom antibody optimization is available upon request.
Chromogenic Multiplex IHC
Up to 4 color (plus hematoxylin) chromogenic multiplex panels are available through MAPcore/GPEC. All panels are optimized on the Leica Bond Rx system using a combination of Leica Polymer Refine Detection systems and Biocare Medical IHC reagents. Our current catalog includes red, DAB, black, and green chromogens. Our current catalog of panels has met the rigorous optimization standards for monoplex DAB stains- plus multiplex validation assays designed to ensure the highest possible performance. Custom panels are available upon request.
Leica Cell DIVE
DIVE into the world of cyclic immunofluorescence! Our latest capital equipment can stain over 60 fluorescent antibodies on a single tissue sample. Our current catalog features key cancer and inflammatory pathology markers. We are continuously expanding and refining our selection of optimized antibodies, please contact us for the most up-to-date offerings.
Opal Fluorescence Multiplex IHC
Up to 7 colours/biomarkers (excluding DAPI) Opal tyramide signal amplification immunofluorescence panels are available through MAPcore. Currently, custom panels only are available though MAPcore. Each Opal panel meets the rigorous optimization standards for monoplex DAB stains- plus multiplex validation assays designed to ensure the highest possible performance. MAPcore Opal panels are optimized to be scanned and spectrally unmixed on our Zeiss Axio Scan.Z1 only. We cannot guarantee the same quality standard if the panel is scanned on any other fluorescence microscope.
in-situ Hybridization (RNAscope)
At MAPcore we use automated ACD RNAscope and miRNAscope kits in combination with Leica Polymer Refine Detection systems on the Leica Bond Rx. Our current catalog contains only brightfield- DAB and Red- assays that have been optimized using positive and negative control tissue and pathologist analysis.
nanoString GeoMx® Digital Spatial Profiler
The nanoString GeoMx® Digital Spatial Profiler, is MAPcore’s flagship assay. The GeoMx® DSP assay starts with UV-photocleavable oligo tagged antibodies or RNA oligos to stains the surface of FFPE tissue or TMA section. IF morphology markers also staining the tissue are used to guide the collection of the UV-photocleavable oligo to generate spatially relevant counts of antibody/RNA targets. The current probe catalog available to be run through MAPcore are Protein and RNA assays run through a nanoString nCounter system.
All GeoMx® DSP assays run through MAPcore are verified by quality control workflow. QC’d data is handed off to each researcher as we currently do not offer full data analysis.

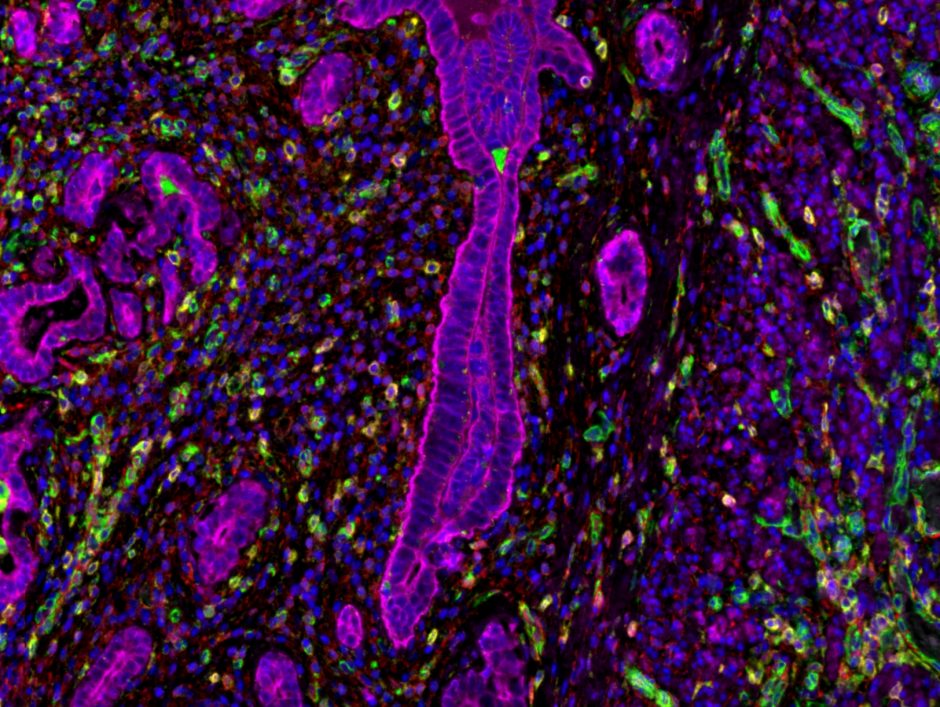
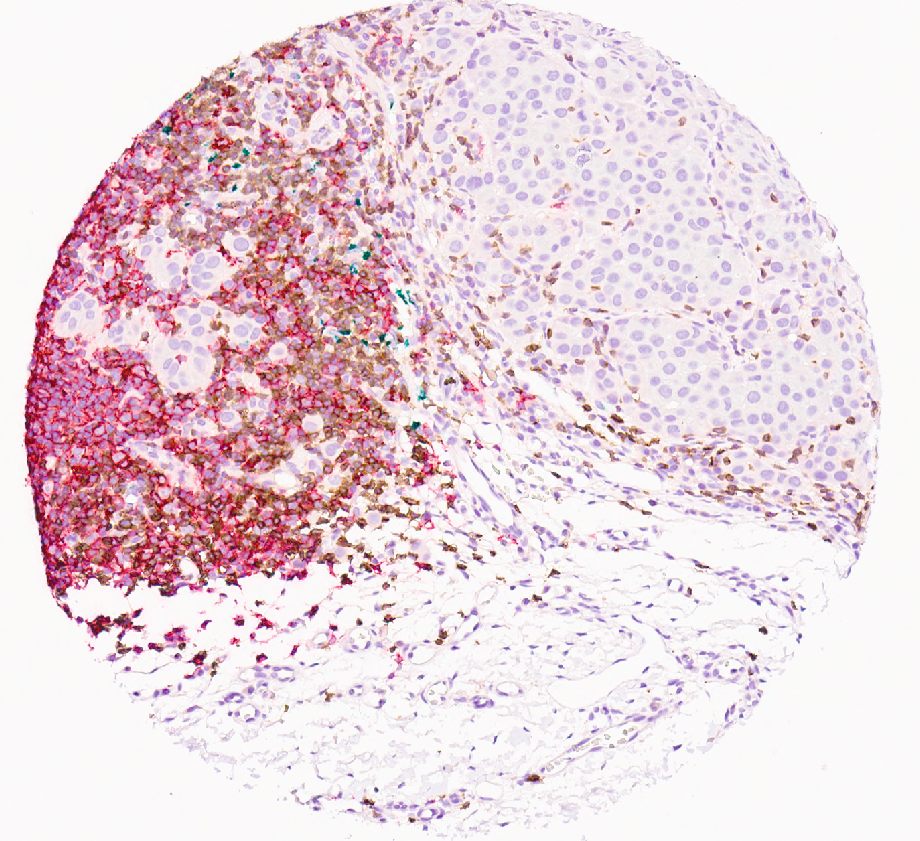
Imaging and Analysis
Brightfield Whole Slide Scanning
Brightfield whole slide scanning is run through the Leica Aperio AT2 microscope. Scan Scope image files are generated by the microscope and can be opened using the freely downloadable ImageScope x64 software.
Fluorescence Whole Slide Scanning
Fluorescence whole slide scanning is run through the Ziess Axio Scan.Z1 microscope. The Axio Scan.Z1 at MAPcore is equipped with the Colibri 7 LED system for fluorophore excitation and an additional 10 emission filters. The Ziess Axio Scan.Z1 generates Carl Zeiss Images (.czi) that can be opened by a number of software’s including: Zeiss Zen Lite, QuPath, Indica Labs Halo, CellProfiler, Python, Fiji & ImageJ, MATLAB and others.
Image Analysis (HALO)
Indica Labs HALO image analysis suite is used by MAPcore to offer analysis of both TMAs and whole slide scans for brightfield and fluorescence images. Modules currently used by MAPcore are:
- Highplex
- Multiplex IHC
- Tissue classifier
- Spatial analysis
- Serial section analysis
- TMA
- FISH-IF
- Halo AI
- Registration transformation export
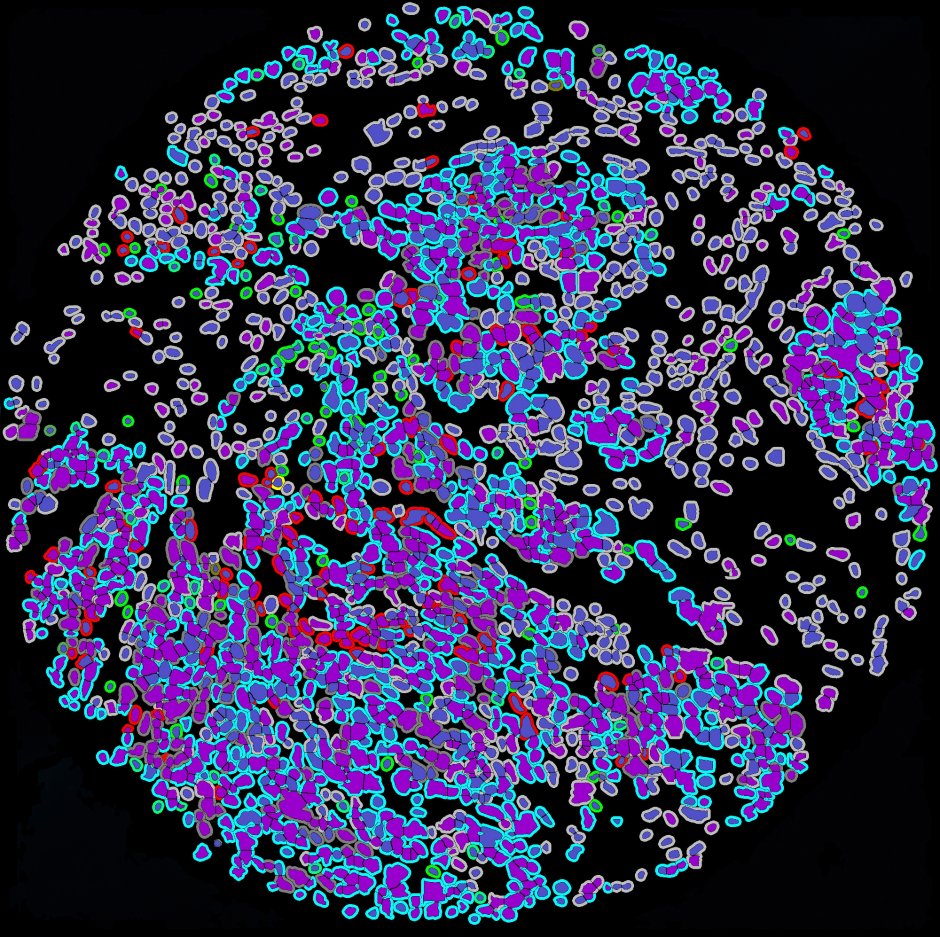


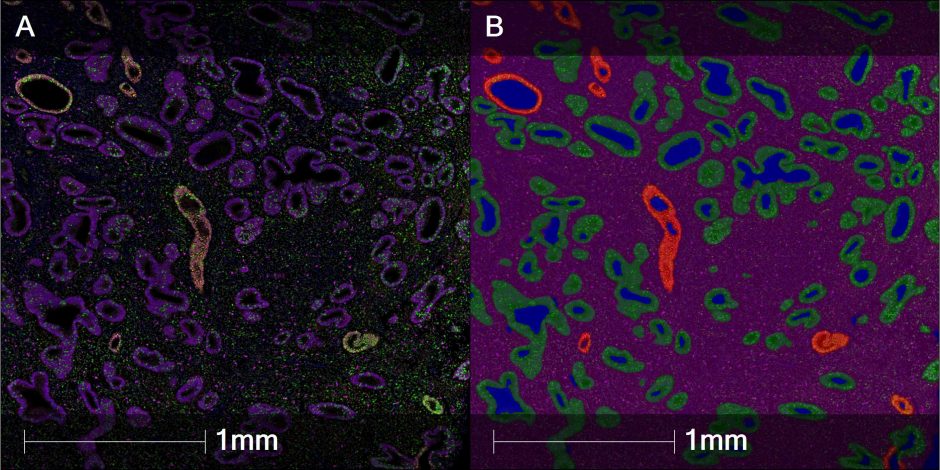
Research Pathologist Assessment
Although interactions with sub-specialty pathologists are best arranged as collaborations, we can provide basic research pathology assessment services including: analysis of tumours, xenografts, and IHC, scoring of TMAs and statistical analysis. We are also open to engaging with users to determine the most appropriate methodology for their projects.
Meet our Research Pathologist!

Dr. Amal El-Naggar is a board-certified pathologist and is currently a senior Research Associate in Dr. David Huntsman’s lab at the BC Cancer Research Centre. Dr. EL-Naggar undertook her medical degree, pathology residency, and her master’s degree in Egypt. She then joined Dr. Poul Sorensen lab at the BC Cancer Research Centre in January 2009 to pursue her PhD at UBC. She enthusiastically focused on conserved cell stress signaling pathways in relation to human pathology and identifying novel targets for the treatment of cancer. Her doctoral studies characterized a previously unrecognized pathway that allows high-risk childhood sarcomas to withstand the stress of hypoxia or cancer drugs. The studies reveal, for the first time, that the highly conserved CSD family member, YB-1, facilitates sarcoma cell invasive and metastatic capacity through direct binding to the HIF1α 5′-UTR secondary structure, likely unwinding it, and therefore enhancing HIF1α mRNA translational efficiency (EL-Naggar et al, Cancer Cell 2015). Following successful completion of her PhD, she continued her training as a Postdoctoral Research Fellow in Dr. Poul Sorensen lab, focusing on targeting specific components of stress signalling to block childhood sarcoma metastasis. During her work with Dr. Sorensen, she integrated her Pathology expertise in the research field, and established a comprehensive xenotransplants-based histopathology and immunohistochemical studies. Then, she joined Dr. huntsman’s lab as a Research Associate. Her work is currently focusing on understanding the critical contribution of stress signaling to lineage commitment and aggressive behaviour of ovarian clear cell cancers, and its pertinent therapeutic targets. Dr. EL-Naggar is collaborating with world’s leading scientists on several research projects focusing on stress response and its link to cancer metastasis, and her contribution to science is manifested in very high-quality publications. She has a wide range of experience from medical and science fields. She has won several prizes and awards for her work such as the poster prize at the BC Cancer Research Day 2018; Early Career Research Program Award 2017; best oral talk award, Ribowest conference 2017 among others. Besides, MSFHR 2017 trainee award. She was also a recipient of Mitacs- Accelerate internship, and a prestigious Egyptian Governmental Scholarship. “The real joy is to pursue a science research career with a Pathologist’s eye”. Dr. EL-Naggar is a mother of a wonderful 13-year-old boy and she genuinely enjoys lots of community volunteering roles.